Products
SunShell Biphenyl
Sunniest Biphenyl
Trifuctional silyl-reagent used
Feature of Biphenyl
The two benzene rings of the biphenyl group are twisted about 45 degree, and the biphenyl group shows different separation from the phenyl group alone.
Even when an aqueous mobile phase containing no organic solvent is used, there is no decrease in retention, and separation with high reproducibility of retention time is achieved.
Greater retention of polar compounds such as caffeine.
Double end-capping
Hexamethyldichlorotrisiloxane
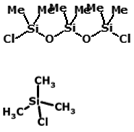
Trimethylchrolosilane
High temperature end-capping
Conversion from silanol groups
to siloxane bond
Low bleeding
High stability
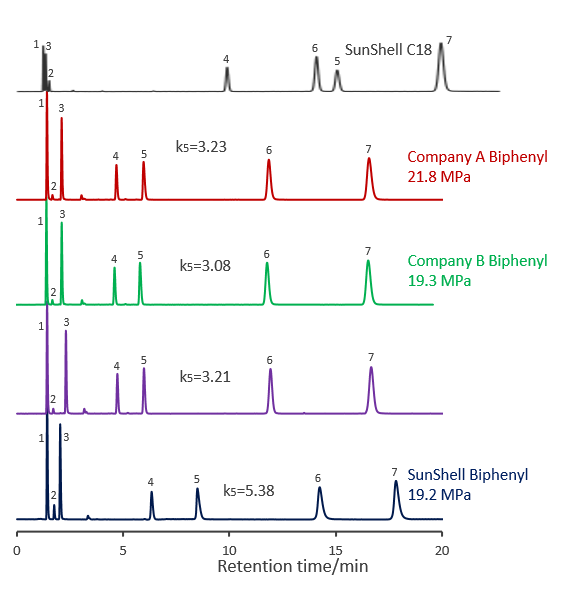
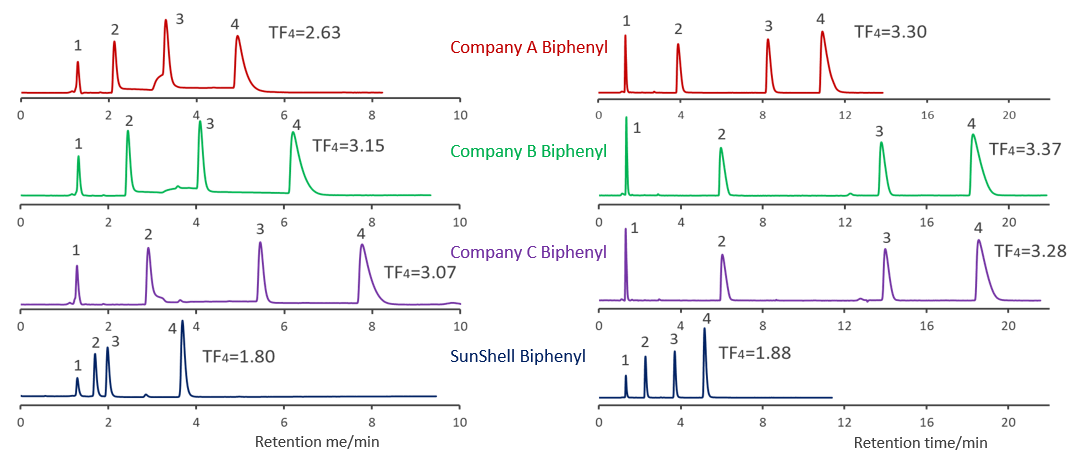
Comparison of core shell Biphenyl columns
Comparison of core shell Biphenyl phases using standard samples
| Hydrogen bond α(Caffeine/Phenol) |
Hydrophobicity α(Amylbenzene/Butylbenzene) |
Steric selectivity α(Triphenylene/o-Terphenyl) |
|
|---|---|---|---|
| SunShell C18 | 0.39 | 1.60 | 1.46 |
| Company A Biphenyl | 2.69 | 1.39 | 1.45 |
| Company B Biphenyl | 2.52 | 1.37 | 1.46 |
| Company C Biphenyl | 3.07 | 1.38 | 1.45 |
| SunShell Biphenyl | 1.82 | 1.44 | 1.28 |
Compared with other company's core shell type Bipjhenyl. Biphenyls from companies A, B and C showed similar separation patterns. It was confirmed that SunShell Biphenyl has a higher retention of amylbenzene No. 5 and a larger separation factor (hydrophobicity in the table) for the difference of one carbon between butylbenzene and amylbenzene, and is more hydrophobic than other company Biphenyl.
The carbon loading is 5% for SunShell Biphenyl and 7% or more for all other Biphenyls, and the high hydrophobicity of SunShell, which has the lowest carbon content, indicates high density end-capping. Moreover, the separation of standard samples is very different for biphenyl and C18.
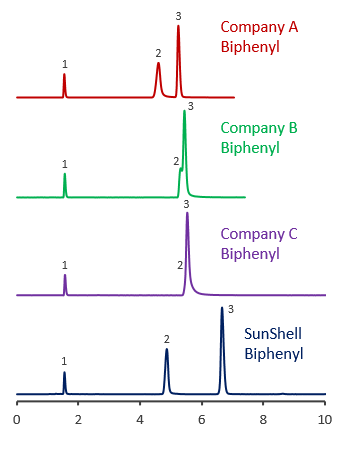
Comparison using pyridine
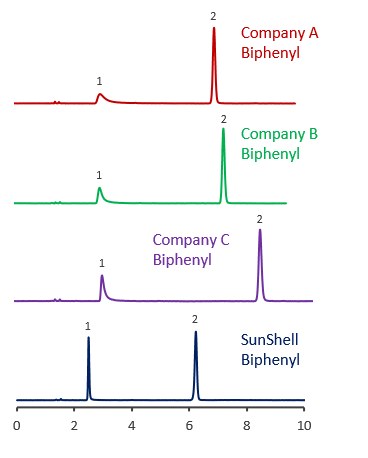
Comparison using oxine
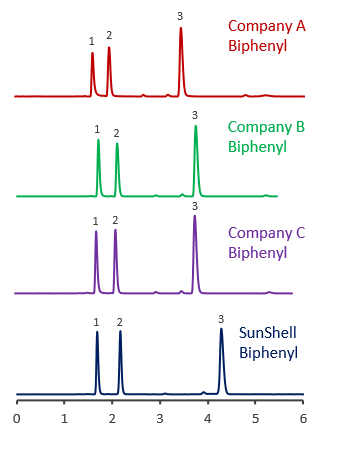
Comparison using formic acid
Comparison of core shell Biphenyl phases using basic compounds

Tailing of basic compounds (amitriptyline) occurs more easily in the acetonitrile/buffer mobile phase than in the methanol/buffer mobile phase. A mixture of acetonitrile and 20 mM phosphate buffer and a mixture of acetonitrile and 10 mM ammonium acetate were used as mobile phase to compare with core shell Biphenyls.
Biphenyl from other companies had a terrible tailing for basic compounds, and it was also confirmed that the retention time was increased, which seems to be influenced by the adsorption to residual silanol groups.
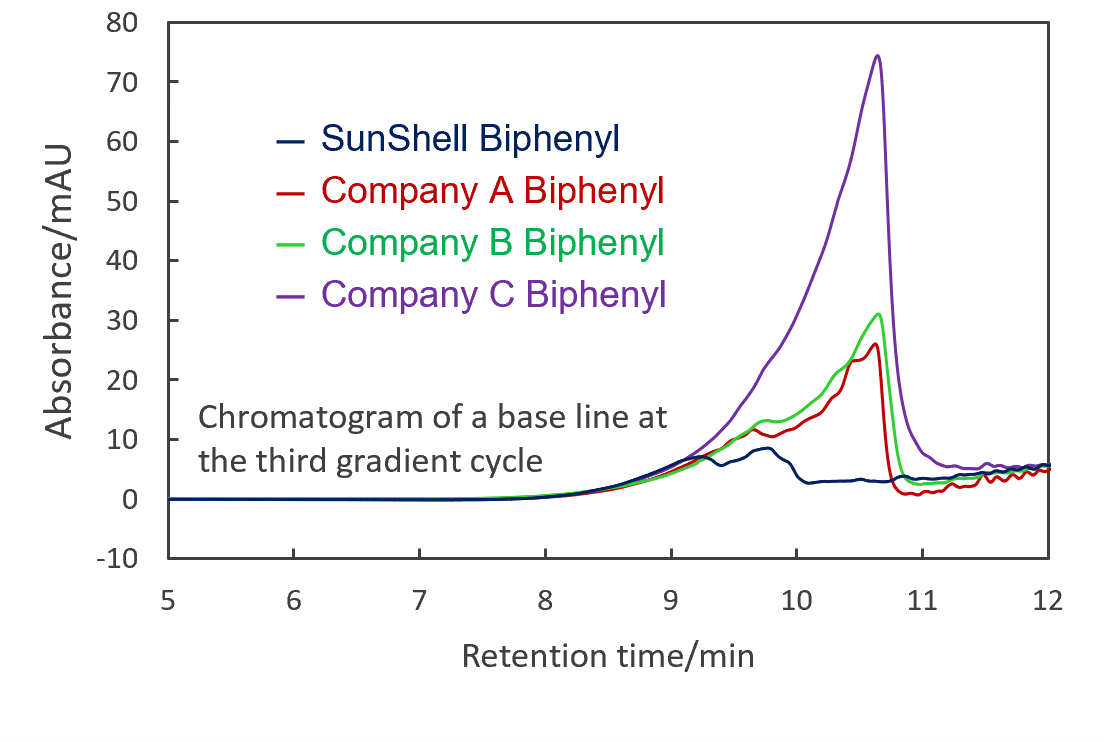
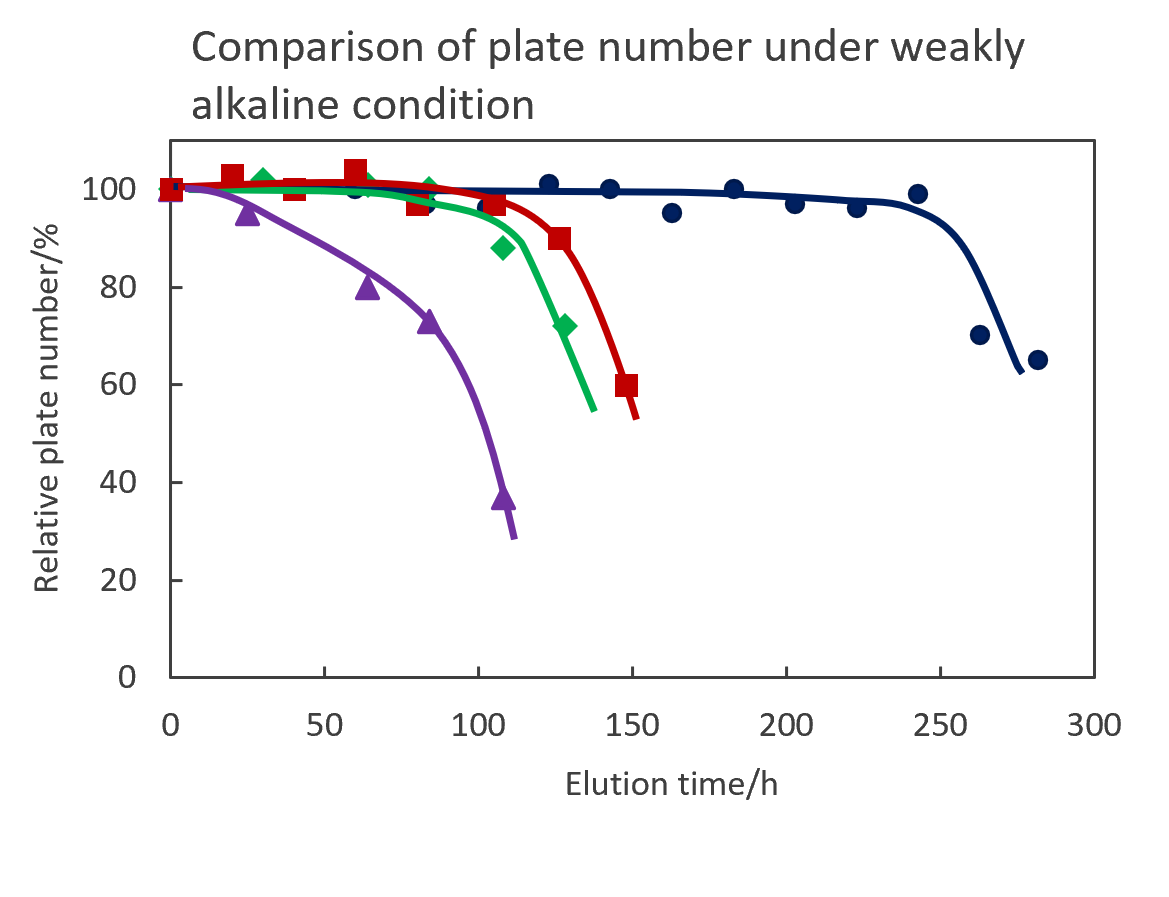
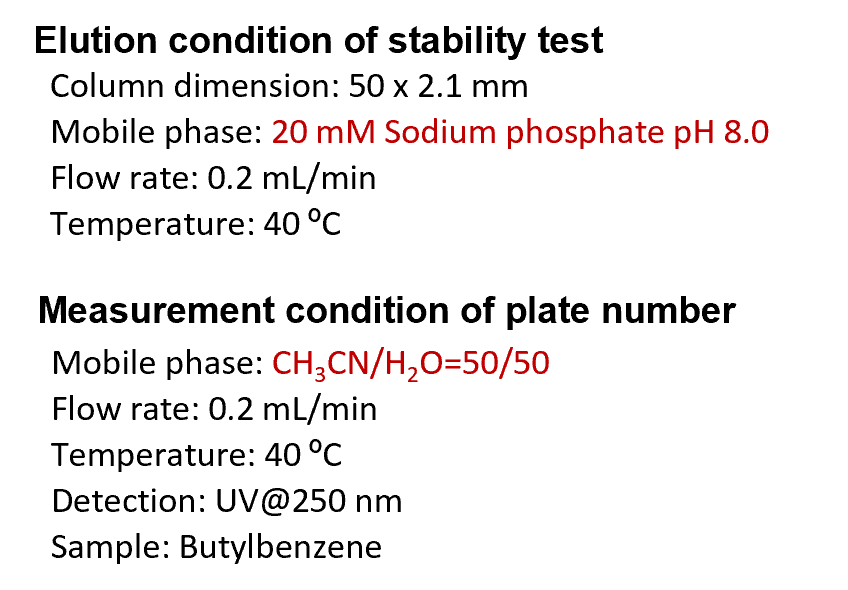
Comparison of stability
Stability under acidic condition compared baseline changes with gradient elution of 1% aqueous phosphate solution and acetonitrile. Biphenyl groups are cut off from the silica surface under acidic condition and elute out of the column as the amount of organic solvent increases. Baseline variability detected cut Biphenyl groups, with SunShell showing the least desorbed and high acid stability. In addition, under weakly alkaline condition, the silica dissolved and the column-in side was dented, so the theoretical plate number of the columns were compared. Since the Biphenyl column has high reproducibility of retention time even in a 100% aqueous mobile phase (see page 6) and is effective for separating highly polar compounds, stability comparison was performed under the condition of pH 8 that does not contain an organic solvent. SunShell is more than twice as durable as other companies' columns.
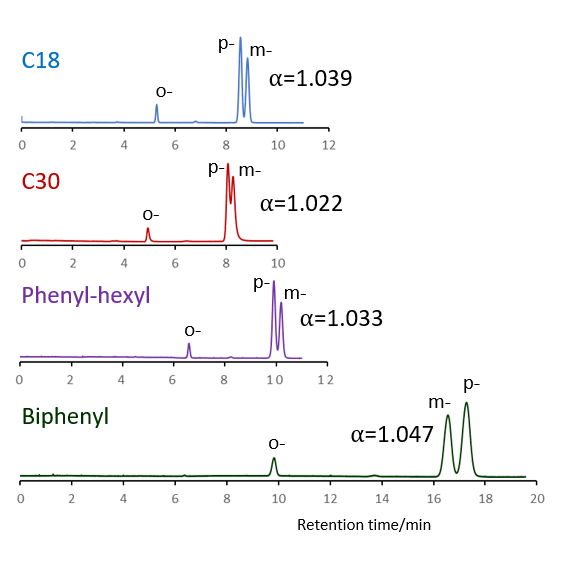
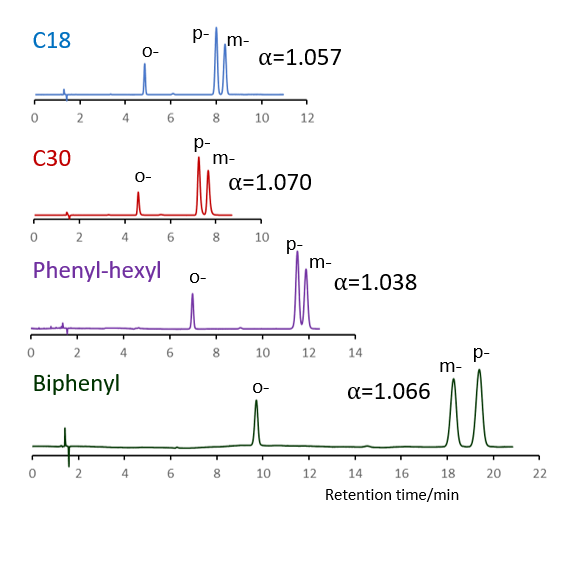
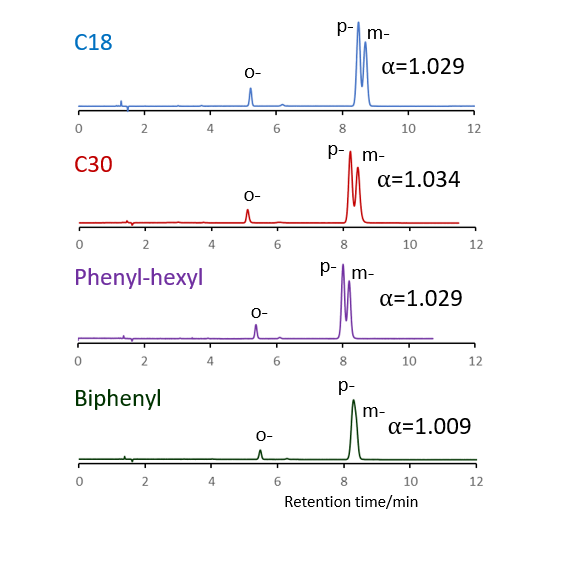
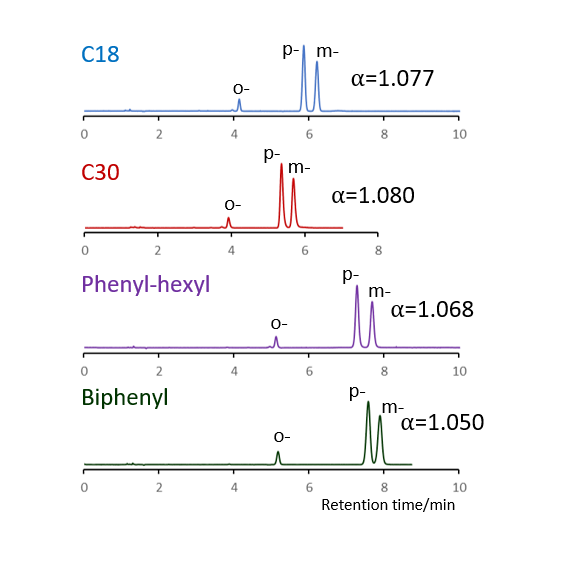
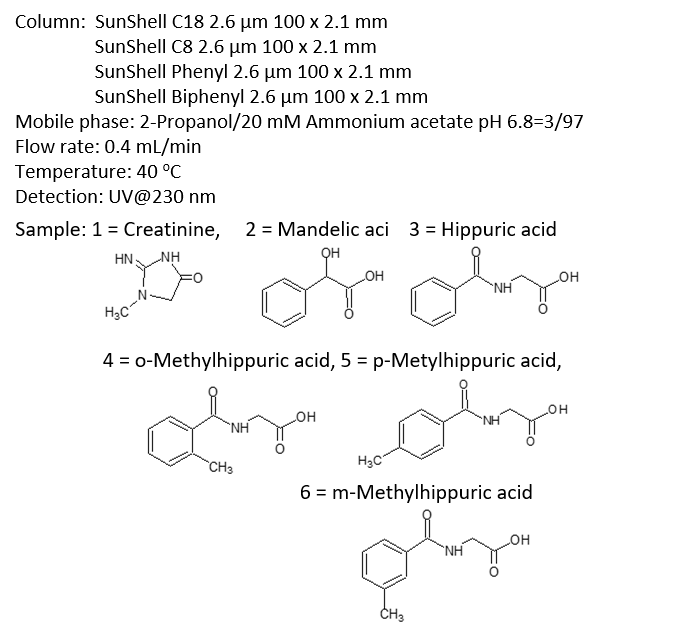
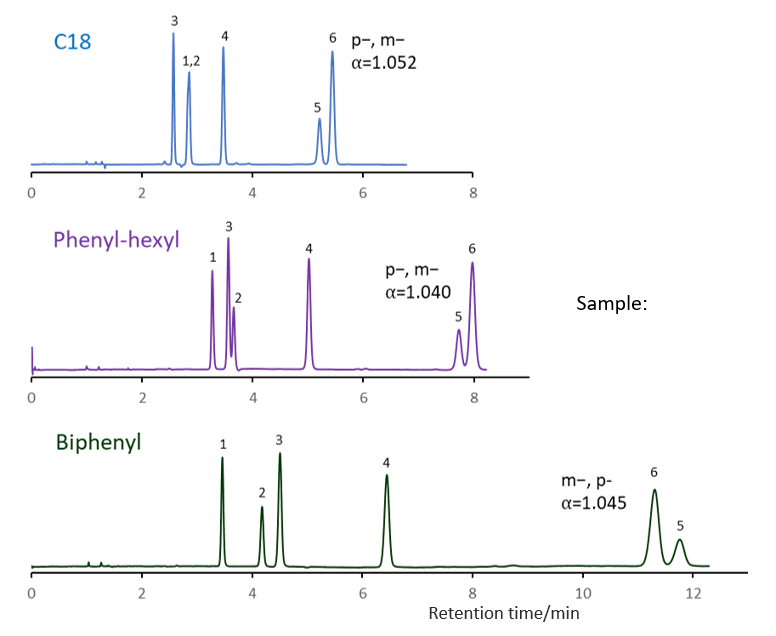
Comparison of stationary phases using isomers of methylhippuric acid

The separation of ortho, meta and para isomers of methylhippuric acid was compared. The stationary phase was C18, C30, Phenyl-hexyl and Biphenyl, and the organic solvent in the mobile phase was methanol, acetonitrile, 2-propanol and tetrahydrofuran.
The separation of meta-methylhippuric acid and para-methyl hippuric acid changed depending on the organic solvent, and the separation was improved in the order of acetonitrile, methanol, 2-propanol, and tetrahydrofuran. Biphenyl showed a particularly high retention when alcohol was used as the organic solvent in the mobile phase, and the elution order of meta and para was reversed from that of other stationary phases. It is considered that this is due to the high hydrogen bonding capacity obtained on comparison of separation of standard samples.
When acetonitrile is used, the π-π interaction between the solute and the stationary phase is weakened by the triple bond of CN in acetonitrile, so it is considered that the characteristics of Biphenyl cannot be fully exhibited. When tetrahydrofuran is used, tetrahydrofuran enters the stationary phase and a mixture of biphenyl group and tetrahydrofuran works as a stationary phase, so it is considered that the separation behavior was different from that when alcohol was used.
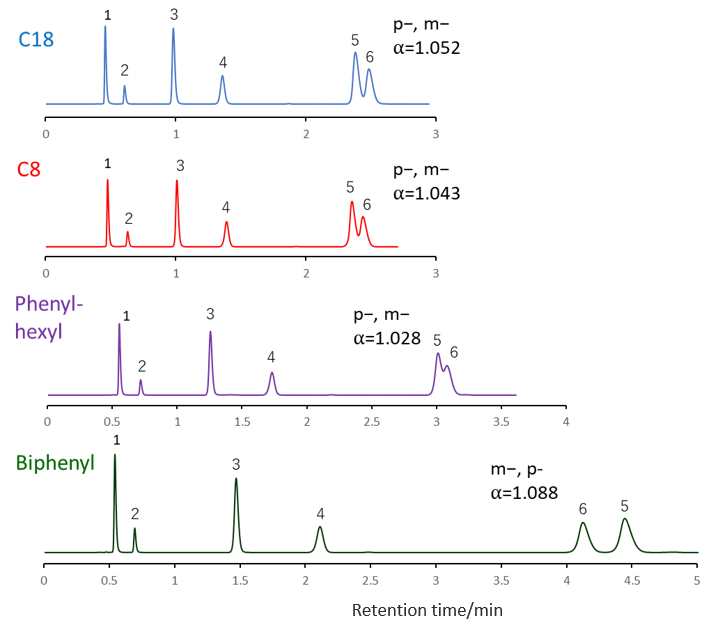
Comparison of stationary phases using creatinine, mandelic acid, hippuric acid and methylhippuric acid
Phosphate buffer was used as the mobile phase for separation of methylhippuric acid isomers on the previous page, but in this comparison of the separation including creatinine, the ammonium acetate buffer mobile phase that can be applied to LC / MS and the mobile phase in which an ion pair reagent were added to a phosphate buffer solution was used.
2-Propanol was used as the organic solvent. Under both mobile phase conditions, Biphenyl showed the highest retention and the best separation. A characteristic separation of Biphenyl is that the elution order of the isomers meta-methylhippuric acid and para-methylhippuric acid is reversed to that of C18, C8 and Phenyl-hexyl
Comparison of stationary phases using nucleic acid bases
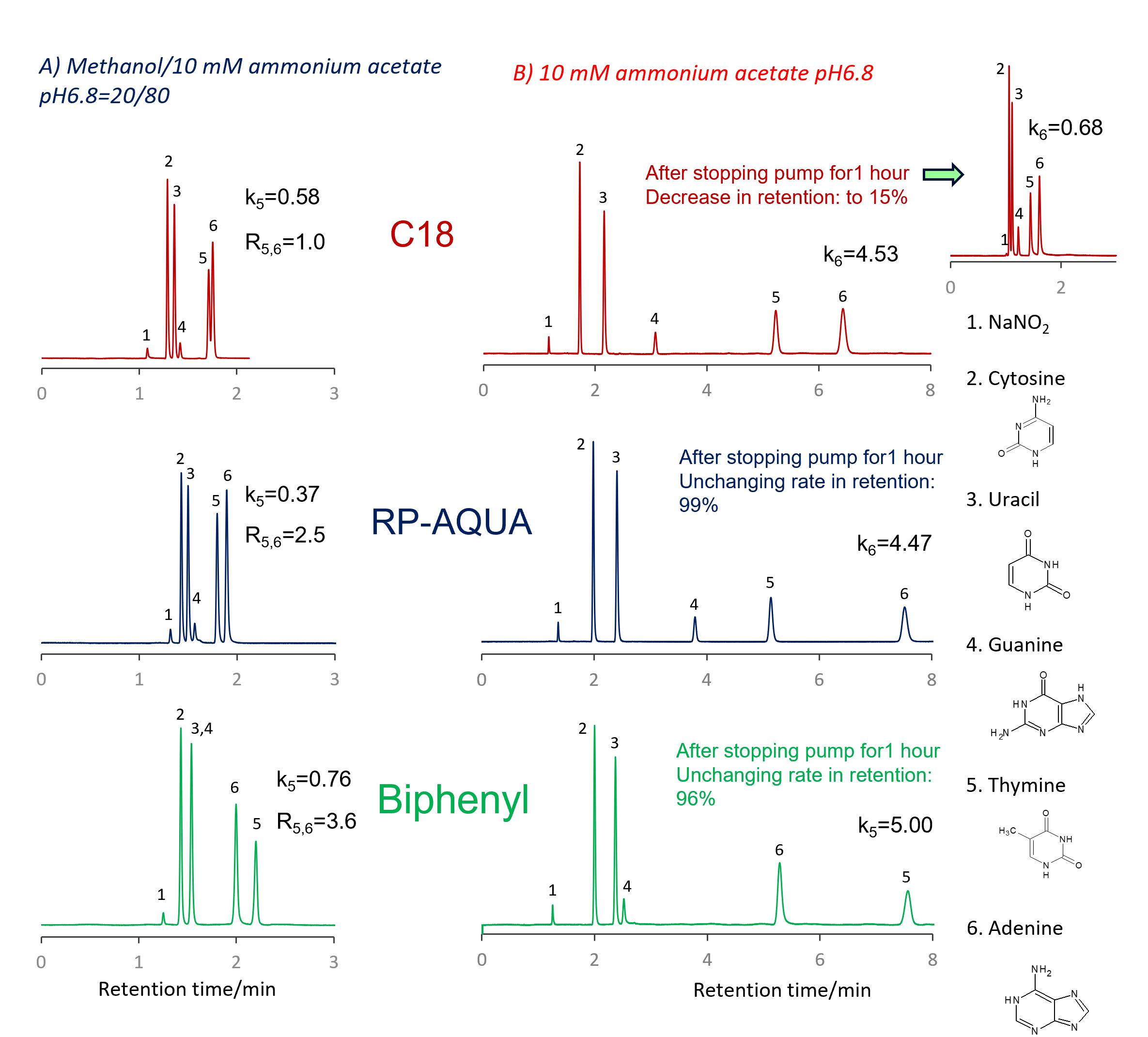

Separation of nucleic acid bases was performed on C18, RP-AQUA and Biphenyl columns. Two mobile phases were used: methanol/10 mM ammonium acetate pH 6.8=20/80 and 10 mM ammonium acetate aqueous solution. Nucleic acid bases can be separated even in the mobile phase containing 20% methanol, but the retention time is shorter.
In comparison of columns used this 20% methanol mobile phase, the retention factor (k5) of thymine at the 5th peak was highest in Biphenyl, and the resolution of the 5th and 6th peaks of thymine and adenine (R5,6) was also highest in Biphenyl.
When an aqueous mobile phase containing no organic solvent was used, the retention factor of thymine eluting at last on Biphenyl was 5.00 and the adenine eluting at last on C18 and RP-AQUA were 4.53 and 4.47, respectively. Biphenyl showed the largest retention factor for the compound eluted at last.
The selectivity of Biphenyl is very different from that of RP-AQUA and C18, such as the order of sample elution. It has also been confirmed that the retention time stability when using an aqueous mobile phase is as high for Biphenyl as for RP-AQUA. The retention factor on C18 decreased to 15% after stopping the flow for 1 hour, confirming low reproducibility in retention.